Most dairy cows experience a period of energy deficit following calving due to increased energy demands of lactation coupled with decreases in nutrient intake. Inability to adapt to this period results in metabolic disease such as ketosis.
This condition is associated with many adverse health and production events, including increased risks of displaced abomasum, metritis, and culling, as well as reduced milk production and reproductive success; all of which are extremely costly to the producer. Despite increasing knowledge about management and nutritional strategies to prevent excessive energy deficit postpartum, ketosis continues to be a problem on dairy farms.
Diagnosis of ketosis is simple and can be performed cow-side with handheld β-hydroxybutyrate (BHB) meters designed to test blood. Cows with BHB concentrations in blood ≥1.2 mmol/L are considered ketotic. Once identified, it is important that these cows are treated to aid in recovery.
Numerous treatment options are available for ketosis, including oral drenching with propylene glycol (PG), a glucose precursor, and administration of intravenous dextrose. Unfortunately, no field trials have been conducted to show the efficacy of dextrose, while there is good evidence that PG is beneficial.
To provide producers and veterinarians evidence-based recommendations, our group aimed to investigate the impact of intravenous dextrose as an adjunct therapy to oral PG by looking at ketosis resolution following treatment, the incidence of subsequent adverse health events, and milk production.
We tested Holstein cows in four New York herds for ketosis once weekly between three and 16 days in milk (DIM) using a handheld BHB meter. Cows with ketosis were randomly assigned to treatment with either oral PG alone for three days (n = 121) or in combination with intravenous dextrose for one day (n = 125) or for three days (n = 127).
All treatments were administered once daily for three consecutive days, and at the same time of day on each farm. Oral PG (300 mL) was administered using an oral drenching gun and intravenous dextrose (50 percent solution, 500 mL) was administered in the jugular vein.
We recorded the number of adverse health events that occurred in cows for the first 60 DIM (sold, died, metritis, displaced abomasum, farm-diagnosed ketosis) and daily milk production for the first 10 weeks of lactation.
In total, we screened 1,249 cows for ketosis with an overall ketosis incidence of 30.1 percent and a median blood BHB at diagnosis of 1.5 mmol/L (Figure 1).
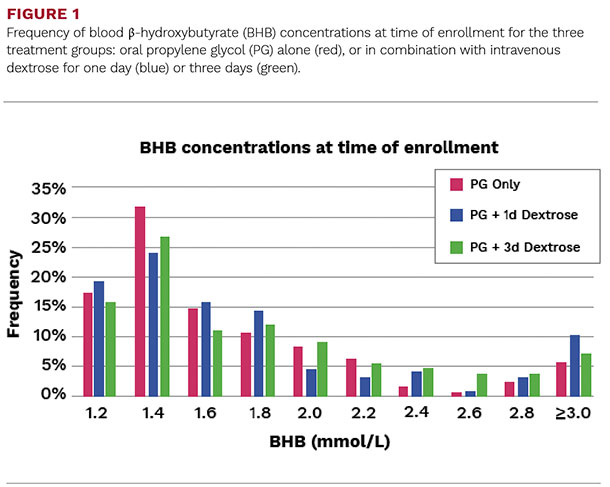
Most cows that developed ketosis were first diagnosed between three to nine DIM (64 percent), whereas only 36 percent had their first positive test between 10 to 16 DIM.
Overall, 9.4 percent of cows had one or more adverse health events during the first 60 DIM, with the most common adverse event being culling or death, followed by metritis, displace abomasum, and farm-diagnosed ketosis. Our results showed that addition of intravenous dextrose for either one or three days to oral PG did not change recovery from ketosis one or two weeks after treatment.
Regardless of treatment group, at one week following treatment, approximately 51.1 percent of cows had recovered, and at two weeks this percentage increased to 58.1 percent. In addition, there was no effect of treatment group on the risk of adverse health events during the first 60 DIM.
Milk production was not different between treatment groups with average daily milk production for the PG only group of 85.1 ± 1.7 lb/d, the PG plus one day of dextrose group of 84.3 ± 1.7 lb/d, and the PG plus three days of dextrose group of 84.7 ± 1.7 lb/d.
In summary, our findings in four herds suggest that intravenous dextrose for one or three days in addition to oral PG for treatment of early postpartum ketosis did not improve recovery, reduce the risk of adverse health events, or improve average daily milk yield over treatment with PG alone.
Ketosis risk, treatment preferences, and occurrence of negative health events vary between herds. Therefore, we suggest that producers discuss the results of this and other studies with their veterinarian to determine the optimal ketosis treatment plan for each individual herd.
This article appeared in PRO-DAIRY’s The Manager in July 2020. To learn more about Cornell CALS PRO-DAIRY program, visit PRO-DAIRY Cornell CALS.









