Composting is a microbial-driven process. Like other living creatures, microbes need the right environment to survive and thrive. For successful composting, it is necessary to provide nutritious microbial “food”, suitable moisture, pH, temperature and oxygen.
What is nutritious microbial “food”?
During composting, microbes break down organic compounds to obtain energy to carry on life processes and acquire nutrients (nitrogen, phosphorous, potassium) to sustain populations. Of the many elements required for microbial decomposition, carbon (C) and nitrogen (N) are the most critical. No other trace nutrients have been found to impede the rate of composting.
Carbon provides both an energy source and the basic building block, making up about 50 percent of the mass of microbial cells. Nitrogen is a crucial component of the protein, nucleic acid, amino acid, enzymes and coenzymes necessary for cell growth and function. The ideal C:N ratio for composting is generally considered to be around 30:1, or 30 parts C for each part N by weight.
As composting proceeds, the C:N ratio gradually decreases from around 30:1 to 10-15:1 for the finished product. This occurs because each time organic compounds are consumed by microorganisms, two-thirds of the C is given off as CO2. The remaining third is incorporated along with N into microbial cells, and then later released for further use once those cells die.
Why start with a C:N ratio around 30:1? At lower ratios than this, N will be supplied in excess and is more likely to be lost as ammonia gas, causing undesirable odors.
Higher ratios, which are not common at dairy composting sites, mean that there is not sufficient N for optimal growth of the microbial populations, so the compost will remain relatively cool and degradation will proceed at a slow rate. In general, materials that are green and moist tend to be high in N, and those that are brown and dry are high in C.
The complexity of the C compounds also affects the rate at which wastes are broken down. The ease with which compounds degrade generally follows the order: carbohydrate, hemicelluloses, cellulose and chitin, and then lignin.
Fruit and vegetable wastes are easily degraded because they contain mostly simple carbohydrates (sugar and starches). In contrast, leaves, stems, nutshells, bark and trees decompose more slowly because they contain cellulose, hemicelluloses and lignin.
Ideally, several raw materials should be mixed together to create the “ideal” range of conditions (C:N ratios: 25-35:1; moisture content: 50 to 60 percent by weight; available O2 concentration: greater than 10 percent; pH: 6.5-8.0; temperature: 130-150˚F; feedstock particle size: less than 1 inch).
However, in the real world this cannot always happen. Fortunately, composting is a forgiving process that can occur over a wide range of conditions, and if you mix materials with an eye to an acceptable moisture content and C:N ratio, you can produce acceptable compost with good management practices. In general, the combination of feedstock quality and compost management will determine the quality of the finished product.
What are the water requirements?
For aerobic composting, the maximum moisture content should be kept at a level that allows for the whole composting process to be aerobic. With this rule, materials containing more fibers, like straw and wood chips, can contain higher moisture content (more than 60 percent) without causing anaerobic conditions.
Composting materials such as paper, grass clippings, soil, and manure with little structural strength should contain less total moisture to prevent anaerobic composting. So the ideal moisture content of the compost pile varies with pile materials.
However, it is commonly believed the optimum moisture content for on-farm composting is between 50 to 60 percent by weight. This should feel moist to the touch when you squeeze a handful of blended materials.
Too low of a moisture content will deprive microbes of water needed for their metabolism, and inhibit their activity, resulting in a slower composting process. Too high of a moisture content means pore spaces in the compost pile will be filled with water rather than air, leading to anaerobic conditions.
Moisture also regulates pile temperature. Drier piles tend to heat up and cool down more rapidly than wetter piles. Materials with different moisture contents can be blended to achieve ideal moisture content. Extra water can be added during the blending process if the original materials are too dry to achieve the ideal moisture content.
What are suitable pH and temperature levels?
The pH, which is a measure of acidity or alkalinity of the compost pile materials, affects the growth and activities of microorganisms, and the fate of N compounds. The optimum pH range is 6.0-7.5 for bacteria and 5.5-8.0 for fungi. When the compost pH exceeds 7.5, gaseous losses of ammonia are more likely to occur.
Certain materials, such as dairy manure and paper processing wastes, can raise pH, while food processing wastes can lower pH. However, pH control for optimum range is difficult and generally not attempted.
The pH varies throughout the pile and the composting process. The pH of the finished compost is generally within a range of 6.5 to 7.5. Measuring pH can be done by sending samples to a laboratory.
Temperatures within compost piles affect microbial growth and activities, and hence the rate at which the raw materials decompose. Higher temperatures result in faster breakdown of organic materials, destroying weed seeds and killing pathogens. However, excessively high temperatures (greater than 160˚F) can inhibit microbial activity.
As shown in Figure 1, mesophiles (moderate-temperature [75-105˚F] organisms) begin the process.
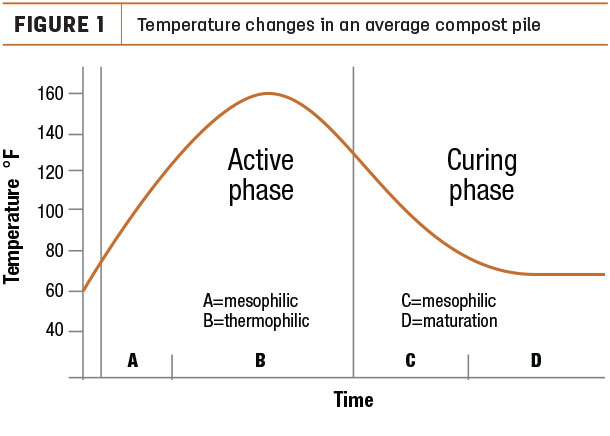 Thermophiles (high-temperature [105-160˚F] organisms) take over during the active phase, and then mesophiles recolonize the pile during the curing phase. Thermophilic temperatures are most effective and efficient for composting. The optimum temperature range is commonly believed to be within 130-150˚F.
Thermophiles (high-temperature [105-160˚F] organisms) take over during the active phase, and then mesophiles recolonize the pile during the curing phase. Thermophilic temperatures are most effective and efficient for composting. The optimum temperature range is commonly believed to be within 130-150˚F.
Temperature should be frequently monitored using a thermometer and adjusted as needed throughout the composting process. Common methods used for adjusting temperatures are aeration, turning and changing pile moisture contents and pile sizes.
What is the oxygen demand?
Composting can occur in both aerobic and anaerobic environments. However, the most efficient way is aerobic composting. Although the atmosphere contains 21 percent O2, aerobic microbes can survive at O2 concentration as low as 5 percent, while O2 concentrations in piles more than 10 percent are considered optimal.
As microorganisms oxidize C for energy, O2 is used up and CO2 is produced. Without sufficient O2, the process will become anaerobic and produce undesirable odors. As microbial activity increases in the compost pile, more O2 will be consumed.
Composts must be aerated either passively or actively, providing aerobic organisms sufficient O2 as active microbes are essential for successful composting. Turning materials is the most common method of aeration for on-farm composting.
Oxygen monitoring equipment is available, but is expensive. Temperature, odors and moisture are easy to measure and provide a good indication of active decomposition and adequate aeration. A compost pile is not odor-free, but a distinct, foul odor (e.g., rotten eggs) usually means anaerobic conditions have developed.
Compost is the product resulting from the controlled biological decomposition of organic material. Quality of the finished compost depends on feedstock properties and management.
While trying to create ideal conditions, be aware that ideal conditions cannot always happen in the real world. Composting procedures are not absolute, but somewhat forgiving. Trial-and-error practices with closely monitoring your composting processes will usually produce usable compost. ![]()

-
Lide Chen
- Extension Waste Management Engineer
- University of Idaho
- Email Lide Chen

.jpg?t=1687979285&width=640)





