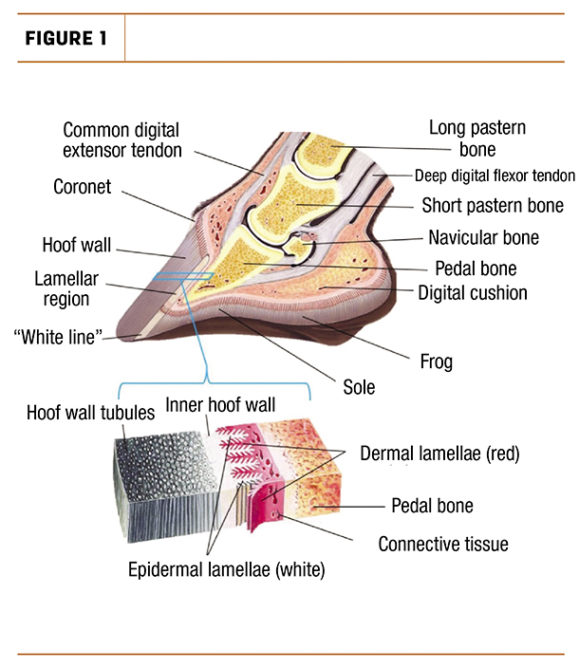
Nothing good can be built on a faulty foundation. On a cow, that foundation is its hooves. Therefore, it’s time to look at what the foundation is comprised of and how the foundation is built. We are still talking cows and hooves, though, right?
Living organisms, unlike architecture, are built organically. That’s why we rely on healthy formation and growth cycles of cellular tissues rather than engineers designing and contractors constructing from industrial resources. Furthermore, environmental factors not only weather the foundation, they impact and contribute to the foundation itself. (And yes, we’re still talking about hooves.)
On average, a mature cow weighs about 1,500 pounds – meaning that theoretically, if each hoof were to bear equal weight, this is 375 pounds per hoof. So just like a bridge, a hoof requires the structure and design to not crumble, provide sound balance and support to withstand weight requirements – except bridges usually aren’t moving around and growing like cows and their hooves. This is precisely why the main protein to provide structural support and strength in bovine hoof composition is so interesting and the main topic of this conversation: keratin.
At a nanoscale level, keratin is a fibrous protein that forms either alpha helical microfilaments (spiral-shaped) that construct tubules, or they layer into beta sheets. Hooves are made up of soft and hard alpha-keratin. The hoof wall and sole are made up of hard alpha-keratin, in which directional tubules overlay each other to provide greater strength and reinforcement. Furthermore, the stratum medium (central epidermal layer) of the hoof wall is known to be the most fracture-resistant biological fibre-reinforced composite – basically “biological cement” (Figure 1).
The main factors influencing the structural integrity (strength and hardness) of hard alpha-keratin are the bonds that hold the spiral helix together (intramolecular bonds) and the bonds between the keratin filaments in the matrix (intermolecular bonds). Furthermore, broken or weak bonds disrupt the matrix integrity, which can lead to weakened soft hooves. There are also scientific studies that found the amount of moisture in these fibrous filaments can impact the hardness. Normal water amount in the hoof wall is at about 17% to 24%, which is the optimal hydration for highest resistance to fracture (proven more tough than fresh bone with fracture stress testing).
There are a few bonds responsible for holding hard alpha-keratin together: ionic, hydrogen and disulphide bonds. The strongest of these molecular structural reinforcements are the disulphide bonds (Figure 2), as they are also linked to the strength and hardness of hard alpha-keratin.
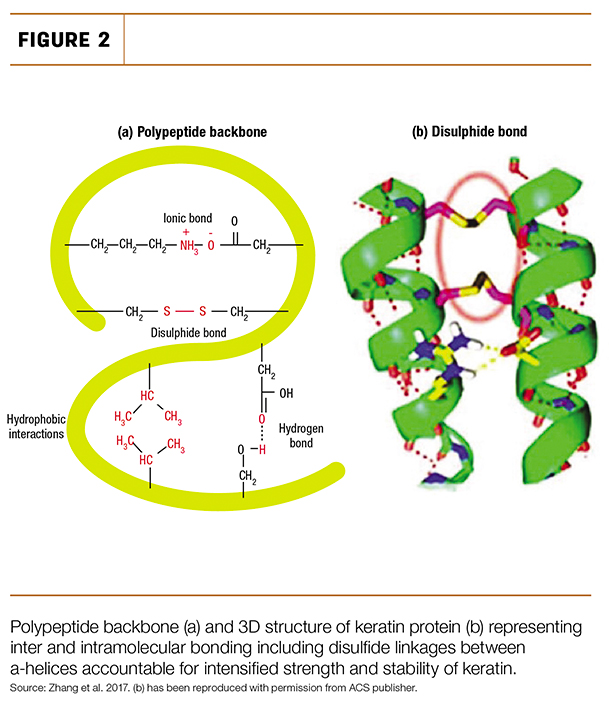
Most interesting is that this particular bond relies on both copper and zinc to form, since these minerals are required to activate enzyme activity at different steps in the formation of these disulphide bonds.
Additionally, there are a few factors to consider that can impact hooves, in relation to the structural integrity and the formation of hard alpha-keratin: bio-resource availability of energy, vitamins and minerals; environmental and housing conditions (tiestall versus freestall, hardness of floor/concrete, free-roaming on pasture, wetness, exposure to harsh chemicals that disrupt keratin formation, etc.); and genetics. Lastly, keratin can modify itself after formation in response to environmental factors, assemble and reassemble microfilaments, interact/signal with other epidermal cells and has a role in vesicle transport.
Research has shown that increased amounts of zinc and copper have a correlation to hoof hardness. Yet neither copper or zinc is biologically produced by the animal; therefore, hooves rely on getting copper and zinc through digestive intake (nutrition) or absorption into fibrous microfilaments topically (footbathing or hoof gel).
As we can now deduct that a good foundation for a cow relies on the structural integrity of the hard alpha-keratin in their hooves, the understanding of common hoof care practices on-farm begins to make more sense. Footbathing, hoof trimming and nutrition all understandably have an important role that can be examined from the context of keratin.
Since copper and zinc are actively involved in the formation of hard keratin in hooves, these products (in sulphate forms) traditionally have been used for over a century in footbaths; however, it is not until more recent years we have fully understood the reasoning behind how these key minerals partake in keratinization. The absorption ability of hoof horn classifies keratin as fibrous, with an amorphous absorbent end, another end as crystalline and insoluble. Since keratin is fibrous, it will absorb water and minerals (including copper and zinc). A study reports that copper sulphate penetrates hooves at a rate of 0.05 to 0.38 millimeter per hour and zinc sulphate at a rate of 0.06 to 0.58 millimeter per hour.
Copper is the activator for the enzyme thiol oxidase, which catalyzes the disulfide bond formation between cys residues of keratin filaments. Zinc is required for several processes to make keratinocytes, including activation of different enzymes and making zinc-finger proteins required to make keratin filaments. Therefore, it is recommended to use hoof care products that contain both copper and zinc sulphate with dairy cattle.