Bacteriological culture and intramammary infusion are two very powerful and important tools of an effective mastitis control program. They have been used for several decades and, when coupled with good management practices, they have helped to produce better-quality milk. Improper supervision and lack of training in this area can make these important tools useless and cost the dairy lots of money.
A successful dairyman in the Northwest called me after reading a very interesting article about the use of extended antibiotic therapy to treat fresh heifers infected with Staphylococcus aureus (SA). He asked for my opinion about the practical possibilities of treating these animals because the number of fresh animals affected with SA mastitis at his farm seemed a little higher than usual.

It had increased from a 2 percent level to over 5 percent on his operation since last year, according to culture results.
He recognized he had tried to treat some animals in the past without much success and too many cows would come back to the hospital with mastitis after a few weeks of being treated.
He got frustrated with the results and quit doing it. Was now the time to start again? To better assist him, I proposed doing a fresh cow and hospital evaluation and sanitation assessment.
I got to the farm about 15 minutes before they started milking the hospital pen. My first observation was that the fresh cows were milked with the hospital cows.
After looking at the milking procedures and the way the milkers were handling the cows, it was time to take hospital and fresh cow samples and at the same time evaluate the intramammary therapy. I will concentrate on the sampling technique and intramammary infusion part of the evaluation, since it was critical for my recommendations.
The correct identification of the affected animal is the first step you need to take if you decide to start treating animals affected with SA. Finding the pathogen in cows that are truly infected is the base for the program.
With some new intramammary drugs, if you do everything right, you can expect to see 80 percent cure rates in fresh heifers affected with SA. The success rate decreases as days in milk and days since infection get higher and also as lactation number increases.

Can sampling technique affect the infection rate?
Absolutely! When taking samples from fresh animals for contagious pathogen screening (usually Staph. aureus and Mycoplasma control programs), it is important that we have a clean representative sample from each quarter.
When all four quarters are stripped into the same vial, we have a composite sample from a cow.
I saw one of the milkers taking a “composite sample” without gloves and dirty hands. When sampling fresh cows, he was taking samples from one or two quarters only. He just wanted to fill up the vial with milk.
This practice may reduce the number of known infected animals in the herd, because he probably missed some positive quarters from infected cows.
Gloves should be worn by the milkers all the time, with no exceptions. The rough surface of the skin doesn’t allow complete disinfection, while gloves provide a smooth surface that is easily rinsed and disinfected.

I like the use of the milker sleeves for the same reason. The gloves and the milker sleeves should be rinsed or changed as often as necessary during milking when they get dirty or break.
Another important factor to consider when taking samples is cross-contamination.
If a cow is infected and we don’t change gloves or rinse our hands between cows, we may contaminate the following samples with milk from the infected cow, therefore increasing the number of healthy animals that will be misdiagnosed and unnecessarily treated.
In this case, training the employees so they know why, how and what they’re doing is a critical part of the mastitis control program.
Disinfection of teats, especially teat ends, with alcohol pads or towels before sampling fresh cows or new cows with mastitis is very important to avoid sample contamination. Do not overflow the pad with alcohol to clean the teats. If the alcohol drips on the sample tube, it will kill the mastitis-producing bacteria we want to isolate, increasing false negative results.
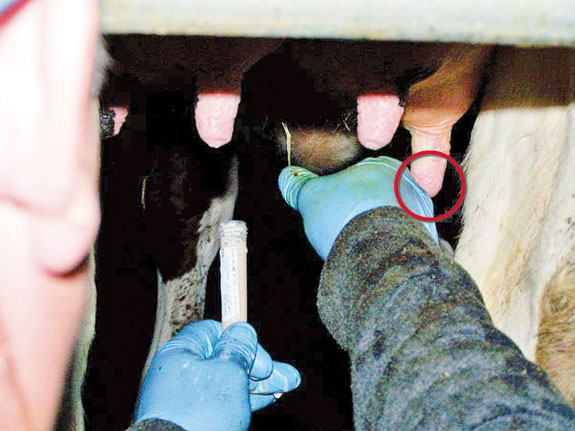
Start with a clean alcohol pad or towel and clean the teat end thoroughly. Look at the pad and, if it still looks dirty, use a new pad until it’s clean. Use a different alcohol pad per quarter sampled.
When sampling all quarters of a fresh cow, start by cleaning the furthest from you and continue with the closest ones.
When taking the sample, start from the closest quarters and finish with the furthest ones. That will help prevent recontamination of teats by the milkers’ arms after cleaning.
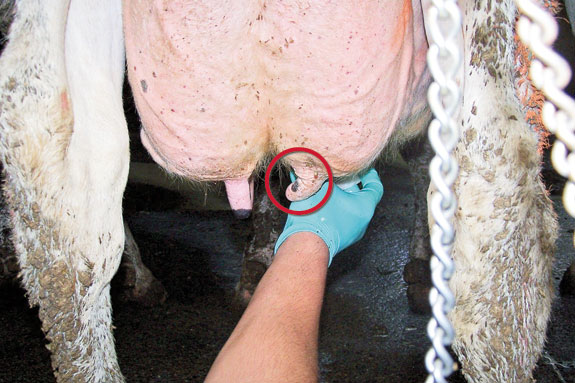
Part of a good sample technique requires that we do not touch the cap or the inside of the tube to avoid contamination ( see pictures ). Leaving the vial opened for extended periods of time should be avoided.
Also try not to position the sample vial directly under the teat while taking the sample to avoid loose debris or dirt falling into the tube. I suggest holding the vial at a 45-degree angle while filling it with one or two squirts from each teat, without touching the teat with the open sample vial.
Intramammary infusion
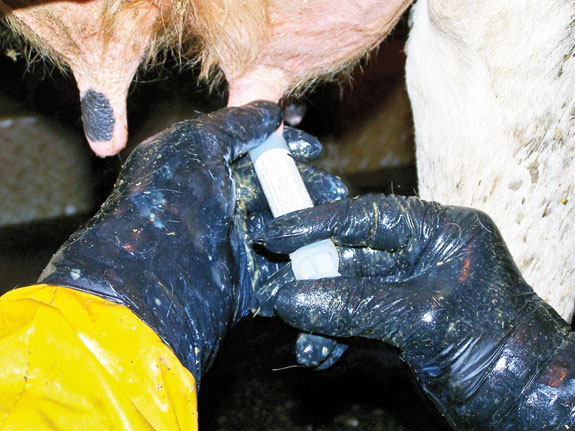
The milkers at this farm were not using any disinfectant to clean teat ends before intramammary treatment. They were introducing the whole tip of the mastitis tube in the teat end while treating. The same disinfection principle we used when sampling quarters applies while treating them.
Mastitis is usually caused by one pathogen. Every time we use a mastitis tube, there’s a risk of introducing new bacteria into the teat canal with the antibiotic if we are not clean during the procedure.
Disinfection of the teat end before treating is crucial to kill most of these contaminant bacteria. These bacteria are in the skin surface or at the teat end waiting to enter the teat when the cannula of the mastitis tube is inserted.
Use a different alcohol pad to clean each quarter treated. Most mastitis tubes come with individual-use alcohol towels that may be hard to unfold sometimes.
I usually recommend the use of Sani-wipes or similar towels to clean the teat ends. They are bigger than individual pre-packaged towels and come in buckets of about 500. These towels are a practical tool to speed up the treatments when you have too many cows in the hospital.
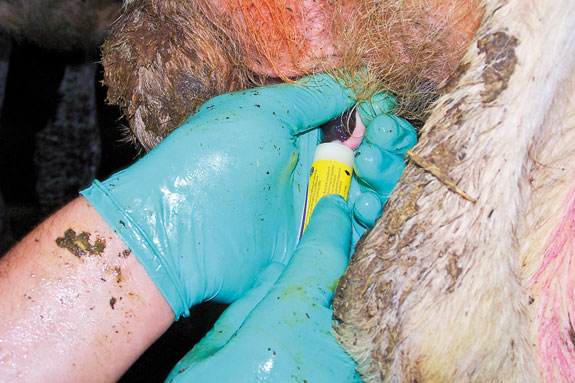
When treating an infected quarter, don’t insert the whole length of the cannula. Introduce only the tip of the mastitis tube (partial insertion) to minimize new pathogen introduction into the teat canal.
Make sure your hands are as clean as possible while treating cows. After treating a mastitis cow I recommend changing gloves or rinsing them thoroughly with water and disinfectant.
The two most commonly used disinfectants in the field are iodine and chlorine. A simple way to have available disinfection is by injecting these disinfectants into the drop hoses in the parlor.
I recommend dialing the pumps so we have about 40 ppm for the iodine and 200 ppm for the chlorine. After treatment, cover the whole teat as soon as possible with a good quality post-dip.
When the dairyman saw my report, we scheduled a milker training session to address general parlor sanitation, milking procedures and also to review sample and treatment techniques. I emphasized the requirement of a non-contaminated sample and aseptic treatment technique to be successful with the new program.
I suggested milking the fresh cows separate from the hospital cows. These transition animals have a suppressed immune system around calving and are more prone to any infection, including mastitis.
We introduced iodine in his drop hoses for disinfection at around 40 ppm. A month after our meeting we regrouped with the owner and the milkers to share the results of this new program.
The analysis showed the new cases of mastitis caused by Staph. aureus in his fresh heifers had dropped to about 2 percent. We also saw a reduction in cows with repeated cases of mastitis coming back to the hospital to be re-treated.
Learning about sanitation and good treatment technique saved the dairy thousands of dollars per year in unnecessary treatments and reduced the number of chronic cows with mastitis. Explain these basic procedures to your hospital manager and teach your employees about sanitation, and you’ll be able to do the same. PD
PHOTOS
DON’TS of hospital parlor mastitis identification and treatments. “I saw one of the milkers taking a ‘composite sample’ without gloves and dirty hands. When sampling fresh cows, he was taking samples from one or two quarters only. He just wanted to fill up the vial with milk.” Photos courtesy Damian Lettieri.

Damian Lettieri
Veterinarian
Udder Health Systems
damian@udderhealth.com









